In this article we will discuss about the Giant Freshwater Prawn (Macrobrachium Rosenbergii):- 1. Distribution of Giant Freshwater Prawn 2. Biochemical Composition of Giant Freshwater Prawn 3. Habit and Habitat 4. Biology and Life History 5. Diseases 6. Culture Practices.
Distribution of Giant Freshwater Prawn:
The giant freshwater prawn (Fig. 8.4), Macrobrachium rosenbergii, is economically important, as it is a fast growing delicious species with considerable export potential. The males and females attain a size of about 25 cm and 15 cm, respectively. Although the adults of this prawn thrives in freshwater and brackish water areas, its larval rearing requires water of about 12 ppt salinity.
This requirement of larvae has caused immense problem for hatchery and grow-out stages of culture. However, to some extent it has been overcome by the use of sea water or brine, trucked in from the nearest source or by the use of artificial sea water.
Macrobrachium rosenbergii is widely distributed in South East Asia besides the Indian subcontinent. It is either reared alone (monoculture) or introduced as a component in carp culture system. One of the suitability of culture of this species is that, the adults are omnivorous and feed on a variety of foods of plant and animal origin.
A major constraint of large scale commercial culture of this giant freshwater prawn is the non-availability of seed, as this is not abundant in nature.
Availability of quality seed, suitable feed and obtaining optimum sustainable yield without environmental degradation are high technological inputs that have been given priority in the past two decades, for farmers to undertake large scale commercial aquaculture ventures in the country.
The semi- intensive culture technology of this giant prawn has been standardised and encouraged for culture as farmers can obtain optimum sustainable yield without environmental degradation.
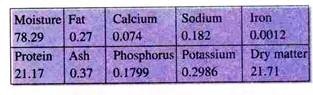
Biochemical Composition of Giant Freshwater Prawn:
Biochemical compositions (%) of adult giant fresh water prawn are as follows:
Habit and Habitat of Giant Freshwater Prawn:
M. rosenbergii is present in rivers, lakes and estuaries of India. It is found distributed from lower to upper reaches of rivers where there is tidal influence. This species prefers shallow and muddy environment. The ecological limits in distribution are water temperature of 25-34°C and salinity 0-20 ppt.
M. rosenbergii is nocturnal in habit. During the day time it hides in shades and shelters. It is sluggish, but shows territorial behaviour. It is pugnacious in nature, but shows less cannibalistic tendency.
Biology and Life History of Giant Freshwater Prawn:
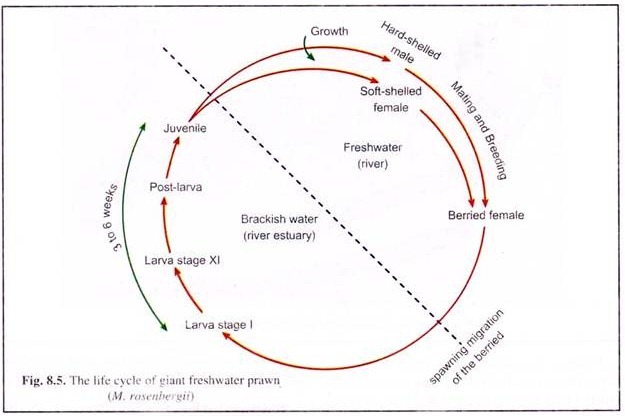
The life cycle of the giant freshwater prawn M. rosenbergii is given in in Figure 8.5.
(a) Food and Feeding Habits:
The giant freshwater prawn is omnivorous, bottom feeder and more importantly it responds positively to supplementary feeds. When it is extremely hungry it resorts to cannibalistic activity.
(b) Growth and Maturity:
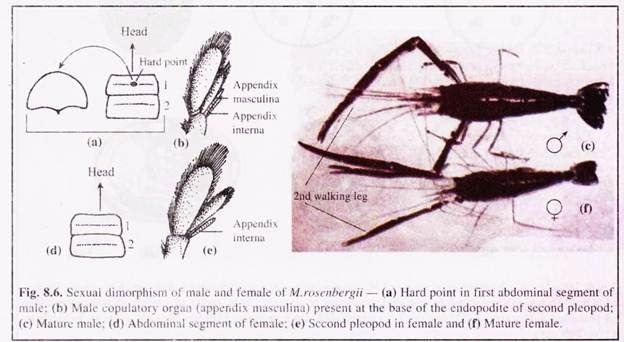
The growth rate of this prawn is fast, showing sexually dimorphic pattern. The males grow faster with heterogenous growth rate. In five months, it attains average growth of 40-50 gm under scientific culture. In the size group of 40-60 gm. the tail of both -the sexes constitute about 55% of the total weight.
This prawn attains first maturity when it is 136- 150 mm in length, weighing about 35-40 gm, within 4-6 months under captivity. In a sexually ripe female prawn, the fully developed ovary is an orange-coloured mass.
(c) Moulting:
M. rosenbergii stops feeding when moulting time gets nearer. At this time, it becomes sluggish, its eye colour becomes dull and the shadow of newly- formed shell appears in the rostrum. Moulting gets completed within ten minutes.
The frequency of moulting is irregular and is dependent upon several factors, such as nutrition, age and stage of sexual maturity. Generally the interval of moulting is 20 to 40 days. The shell gets hardened within 2-6 hours.
(d) Mating and Spawing:
The female prawns before mating moults and this moulting is termed as pre-mating or pre-spawning moult. Mating can be observed by introducing a mature male and a sexually ripe female (3 to 6 hours after pre-mating moult) in an aquarium tank containing freshwater. The female is held strongly by the male between its chelate legs.
The male then cleans the ventral part of the thoracic shell, which takes 10 to 15 minutes and then places the female ventral side up. It then presses from above and ejects the sperm through its genital pores in one gelatinous mass on the ventral thoracic region of the female. The male then releases its grip and the female swims away.
Six to twenty hours after mating, the female lays eggs and the whole batch of eggs is laid in 20 minutes. The eggs at the time of extrusion are considered to be fertilized and they are held within the pleopods in bundles with the help of extremely thin elastic membrane secreted by the ovigerous setae.
ADVERTISEMENTS:
Unmated ripe females also lay eggs within 24 hours after the pre-mating moult. As the eggs are not fertilised, they drop off in 2 or 3 days.
The size of the egg is about 0.6 mm to 0.7 mm in diameter and weighs about 0.1 mg. It is slightly oval in shape. A medium sized prawn of 180 cm length and 80 gm weight lays about 60,000 eggs.
Larger ones, depending upon its size, lay about 1,00,000 to 1,70.000 eggs. In tanks, mature female prawns, spawns twice in five months. In natural condition, however, 3 to 4 spawning may occur in one year. The species have an average fecundity of 500 larvae/gm body weight of mother prawn.
(e) Embryonic Development:
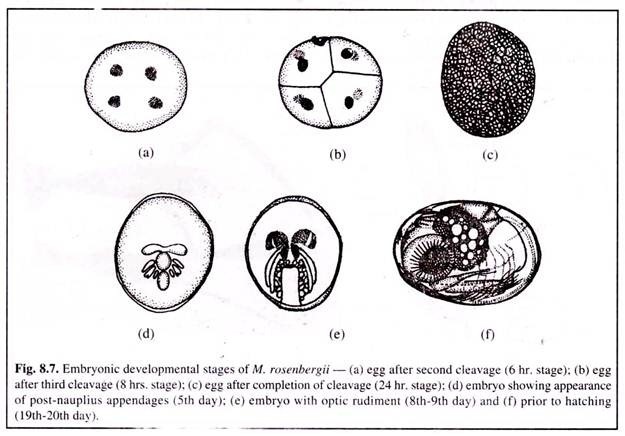
Berried females actively migrate to estuarine regions for the purpose of hatching their young. The incubation period is 18-21 days and requires a temperature of 28-30°C. The eggs are carried by the females during this period. They aerate the eggs by beating their pleopods.
Fertilised eggs have a homogenous granulation and are bright orange in colour. Cell division begins 4 hours after fertilisation. Development continues (Fig. 8.7) and the bright orange colour of the eggs begins to fade to a pale-grey and finally darken to slate-grey prior to hatching.
On the 19th or 20th day after fertilisation the larva hatches out. During the time of hatching, the mother prawn vibrates her pleopods at intervals to disperse the eggs.
(f) Larval Development:
The newly hatched larvae (Zoea) are planktonic and attracted to light (not bright illumination). They require brackish water for their survival and metamorphosis. In nature larvae hatch both in freshwater and brackish water. Those in freshwater are carried to estuaries by water currents, otherwise in freshwater they will die in 4 to 5 days.
The larvae are active swimmers. They swim tail first, ventral side upwards at an oblique angle. Early larvae are gregarious in habit. They swim together and in 10 days’ time the gregariousness gradually disappears. Larvae are omniovorous, feeding on zoo- and phytoplankton and other organic foods like bits of flesh, prawns, molluscs as well as grains, seeds, fruits, etc. Liquid food is not taken.
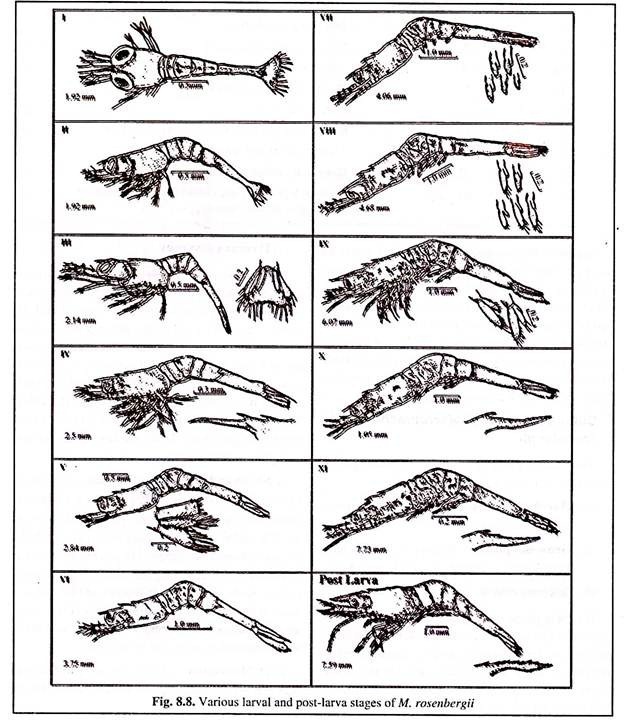
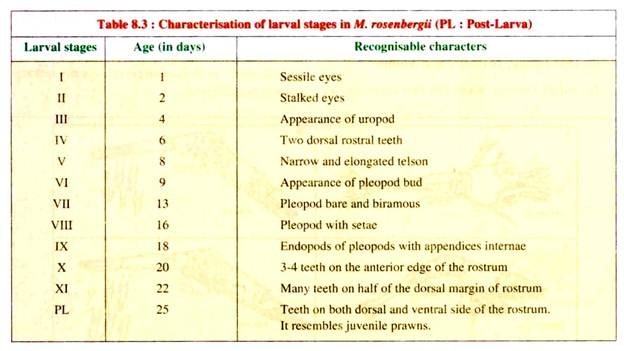
Larvae passes through eleven moults and the completion of larval cycle takes about 30 days depending upon the temperature and feeding conditions. The different larval stages are given in Fig. 8.8 and their recognised characters are depicted in Table 8.3.
The total length of different larval stages ranges from 1.92 mm (stage I) to 7.73 mm (stage XI), while the newly metamorphosed post- larvae measures 7-8 mm (Fig. 8.8).
(g) Post-Larvae, Juveniles and Adults:
Juveniles emerge when the last stage larvae metamorphose and cease their pelagic life. They sink down and settle at the bottom as crawler and clings to vegetation and submerged objects. They feed on organic matter as well as on small worms, crustaceans and insect larvae.
In nature, the juveniles remain for 1-2 weeks in brackish water and then gradually migrates upstream against the current, into less saline water. During rainy season they migrate into ponds, streams, paddy-fields, etc.
They are generally found to cover 200 km by migration. They then become sexually mature in about nine months. The adult prawn’s body gradually changes from their original transparency to bluish or brownish colour.
Diseases Encountered by Giant Freshwater Prawn:
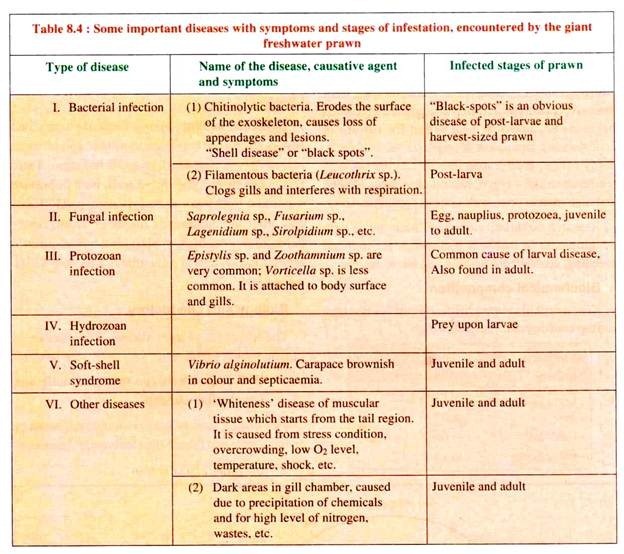
M. rosenbergii, although considered as a freshwater prawn, is not purely a freshwater species as it has to undergo a saline phase in its life cycle. This giant freshwater prawn is susceptible to various types of diseases, particularly during the intermoult period. Some common diseases with symptoms and the infected stages of the prawn are given in Table 8.4.
Culture Practices of Macrobrachium Rosenbergii:
The culture practice of this freshwater prawn comprises two major phases:
(A) Hatchery Phase:
Hatchery phase includes hatchery system, management of broodstock, spawning, hatching and larval collection, larval rearing and acclimatisation to freshwater under controlled conditions in the hatchery. A nursery system may also be introduced before stocking in grow-out ponds.
(a) Hatchery Systems:
Various hatchery systems are in use in commercial farms. The more important ones belong to clear water system, recirculating water system and the more commonly used is the green water system (Fig.8.9). Many hatcheries comprises mating, spawning and larval rearing — all occurring in the same tank.
Larger tanks are generally required for water storage, post-larval rearing and for Artemia hatching. The tanks are generally kept indoor to prevent the water from getting hot during the day of summer, and too cold at winter nights.
(i) Shape and size:
Hatchery tanks are made of various shapes and sizes. They are made of various materials — plastic, fibreglass, reinforced concrete, etc. The shape of the tanks may be circular with flat or conical bottom, rectangular or square, each having its advantages.
The rectangular tanks are more space- saving, with water and air intakes at one end and drain at the other. Circular tanks, on the other hand, are preferred as they are easy to clean and with better water circulation are easier to maintain.
(ii) Colouration:
The tanks are painted with dark colours so that the food can be well-seen by the larvae.
(iii) Aeration:
An aeration system is attached to the bottom of the tank to facilitate proper mixing of the water and even distribution of the food particles and also the larvae. The even distribution of the larvae helps in reducing cannibalism.
(iv) Water management:
Water management procedures of larval culture refer to terms such as green water, clear water and recirculating water systems.
(1) Green water system:
As the name specifies, the green water system (Fig. 8.9) involves the production of phytoplankton culture dominated by Chlorella. To maintain the plankton bloom, a fertiliser mixture of 4 parts urea and I part NPK (Nitrogen: Phosphorus: Potassium = 15: 15 : 15) in tap water, is added to the tank at least once a week.
Production of rotifers is also maintained by the addition of CUSO4 (0.6 ppm) into the tank water. The salinity of the green water should not be more than 12 ppt. For water exchange of larval tanks, only green water of less than three days old is used.
The function of green water is to maintain the water quality in the larval tanks by recycling the waste products of larval metabolism. It provides food for live food organisms such as Artemia. The only drawback in this system is that only a lower stocking density is feasible.
(2) Clear water system:
This system is adopted for different level of larval production, where circular tanks with conical bottoms (Fig. 8.10) or rectangular tanks are used. Chlorination followed by dechlorination, UV light, strong aeration and use of antibiotics are the measures taken to maintain the water quality. In this system high stocking density is feasible, but it requires high management skills.
(3) Recirculating water system:
This system is valuable for water conservation and use of energy. The tank water is re-circulated through a graded sand/gravel filter, attached to mechanical or airlift pumps. The water quality is maintained by involving mechanical and biological filtration, chlorination and dechlorination or the use of ozone. However, commercial application of such a system has not gained much importance in prawn culture.
(b) Management of Broodstock:
One important aspect in hatchery management is the availability of berried prawns over an extended period, probably round the year. For this, it is essential to develop viable broodstock in captivity.
This involves the following steps:
(1) Freshwater ponds are prepared in a manner similar to that practised under carp culture system. It is fertilised with cattle manure. 15 days before stocking of prawn seed.
(2) Post-larvae or early juveniles of prawns are obtained from the market and stocked in the ponds.
(3) The juveniles are fed with high quality ingredients in the form of pellets, which is broadcasted throughout the pond to enable all the prawns to feed. In absence of pelletised feed, supplementary feed of plant and animal origin is given.
(4) Within 5-7 months of culture, prawns attain maturity in freshwater pond depending on temperature, food and ecobiological conditions.
(5) Maturation takes place in freshwater. Berried prawns are collected by means of a cast net from the pond for hatchery purpose.
Berried females can also be obtained from other farmers initially. In such cases they are brought to the farm in open containers with fresh- or brackish water or in case of long distance transportation they are packed under oxygen pressure at the rate of 2 to 5 prawns per bag.
On arrival at the hatchery, the berried prawns are disinfected by placing them in a container containing 0.3 ppm of copper sulphate or 20 ppm of formalin for 30 minutes.
(c) Spawning:
The berried prawns are released into a tank at the rate of 5-10/m: depending upon the size. The tanks are filled up with brackish water of 5 ppt salinity for efficient hatching. The hatchery tank should be cleaned every day with daily exchange of water of identical salinity.
Mating takes place between hard-shelled males and ripe soft-shelled females. The females are soft- shelled as they have just completed their pre-mating moult. The pleopods of the female are vigorously moved to keep the lertilised eggs well-aerated.
The incubation period varies between 18 to 23 days at a temperature of about 28°C. The ovarian eggs of the berried females, even during the incubation period, may start maturing and a second pre-mating moult can occur within a period of about 3 weeks.
Healthy berried female prawns are selected from either natural sources, rearing ponds, brood tanks or aquaria and introduced into the larval tanks. At the time of selection it is important to ensure that all the females have black or grey eggs. This would ensure that all the eggs hatch out within two or three days and the larvae in the tanks will be of the same age.
(d) Hatching and larval collection:
When all the eggs have hatched, the spent females are removed from the larval tank with the help of a coarse mesh dip net. Larval release from the pleopod of the female prawns occurs over a period of four days, with a peak during 24 to 72 hours.
Therefore, the females have to remain in the larval tank for a period of 4 days. The salinity of the water is then raised to about 12 ppt, with an optimum temperature range of 26-31°C. The tank is aerated to maintain oxygen levels nearly to saturation point. In case of clear-water and recirculating system hatcheries, the tanks are not exposed to direct sunlight.
In farms where larvae hatch out in the hatching tank, they are removed and stocked in the larval rearing tank. The larvae after hatching in the hatching tank are attracted to one corner by means of a light and they are siphoned into a container with a known volume of water. The total number of larvae in the container is estimated and then stocked in larval rearing tanks.
(e) Larval rearing:
The newly hatched larvae (zoea) are planktonic and require brackish water for their survival and metamorphosis. They undergo eleven stages of metamorphosis (Fig. 8.8 and Table 8.3). After 16-28 days (depending upon the temperature) the post- larval stage is achieved and the larvae are harvested for stocking in grow-out or nursery ponds.
(i) Water intake and treatment system:
To achieve optimum larval survival and seed production, it is essential that the sea water and freshwater used should be clear and free from pollution. The sea water collected is first stored in cemented tanks and chlorinated with bleaching powder (20 gm/1000 litres of water) to kill all the micro-flora and -fauna.
The water is then allowed to settle for a week and then used for preparation of larval medium. The medium is then passed through 5-10 micron filter bags and introduced into larval rearing tanks. The water from time to time should be checked for suitable hardness (50-100 ppm), iron (below 0.1 ppm) and level of heavy metals.
(ii) Larval stocking:
Larval stocking and rearing can be done following two-phase larval rearing system utilising cylindro-conical tanks in conjunction with the large tanks having flat or U- shaped bottom. This system is suitable for obtaining high survival and production of post-larvae.
In the first phase, early larvae (stages I to V or VI) are reared in cylindro-conical tanks with high density stocking of 500 to 700 larvae per litre.
In the second phase of rearing, the advanced larvae stage of V or VI are collected from the first phase of rearing and stocked in large tanks at the rate of 50 to 80 larvae per litre.
(iii) Larval feed:
(1) Feeding of brine shrimp (Artemia sp.):
Freshly hatched Artemia nauplii are supplied as food, to the prawn larvae in its early stages (stages II to VI). Feeding is done twice a day and for the later stages, once during late evening in combination with prepared feeds.
The cysts of brine shrimp are hatched in a conical tank whose capacity usually ranges between 100 and 500 litres. Hatching instruction of the cysts are normally given by the suppliers.
But certain conditions are required for optimum hatching which is given below:
The Artemia nauplii after hatching are disinfected with 70 ppm of formalin after which they are fed to the prawn larvae.
(2) Prepared feed:
A number of feed ingredients of animal and vegetable origin are combined variously to prepare diets for feeding the larvae. The feed ingredients are mixed together with a little water, in an electric blender or mixer to form a bolus which is then steamed for 30 minutes. It is then cooled and stored in a refrigerator. The refrigerated feed should be utilised within four days.
The ingredients and its composition used for formulating a diet suitable for feeding prawn larvae are given below:
(iv) Method and feeding schedule:
For high survival and metamorphosis, proper feeding level and feed formulation is an important factor. At the 2nd or 3rd day after hatching, feeding of the prawn larvae with brine shrimp larvae is initiated. The Artemia nauplii is given preferably in the evening, so as to ensure the presence of food in the medium all throughout the night.
At later stages of development, the prawn larvae are given prepared feed, sieved through a mesh to obtain suitable particle size. This is given during the day time at an interval of 2-3 hours.
(v) Cleaning of larval rearing tank:
Larval rearing tank should be properly cleaned to remove the excess food particles and metabolic wastes that have settled at the bottom of the tank. This is done by siphoning daily after stopping aeration and preferably in the evening hours, prior to exchange of water and introduction of brine shrimp nauplii.
(vi) Water quality management during larval rearing:
Water quality in hatching deteriorates due to accumulation of water and unutilised feed. So, the main problem in modern hatchery management is the maintenance of good water quality. The optimum water quality parameter to be maintained is given below:
The water quality in larval rearing tank is maintained through exchange of 50% of water every day. Water quality in the larval rearing tank is also maintained by means of recirculation through biological filter using materials such as gravel, activated charcoal, molluscan shell, etc.
Biological filtration is a process of mineralisation of organic nitrogenous compounds, nitrification and denitrification of bacteria, suspended in the water and attached to submerged objects in the filter bed.
(f) Acclimatisation to freshwater:
The post-larvae are acclimatised gradually to freshwater. During this period aeration should be continued and after acclimatisation the larval feed is stopped. The post-larvae are then fed on a variety of food such as small shrimp (Acetes sp.), worms, crushed maize, broken rice, molluscan meat, tapioca, etc.
After a week or two, the post-larvae become suitable for stocking in grow-out ponds. If they are reared for more than two weeks, then over-crowding will take place and the survival rate will decrease.
(g) Introduction of Nursery system of rearing:
A nursery system of rearing may be introduced before stocking in grow-out ponds, where newly metamorphosed post-larvae of Macrobrachium rosenbergii are reared to a juvenile stage (25 mm).
Shallow freshwater ponds of one metre depth and a water area of 200 to 500 m2 are preferably used. The pond bottom should be made of clay or clay- loam. The pond should be prepared by liming and use of fertilisers (cattle manure, urea, single super phosphate), at least 15 days prior to stocking.
The quality of pond water should be maintained and should be free from toxic chemicals and other pollutants. Some important water quality parameters to be maintained for successful rearing are as follows:
The post-larvae are stocked at a density of 50-100/m2 and reared for one to two months, by which through proper feed, they attain a size of 0.5 to 1.0 gm.
Suitable foods given are:
(1) Living aquatic animals—small aquatic worms, insect larvae.
(2) Small pieces of clams, snails, squids, shrimps (Acetes sp.).
(3) Plant material—broken rice, other grains, tapioca, sweet potato, peas, oil cakes, etc.
Formulated feed (pellets) are also recommended at the rate of 20% of post-larval body weight. The feed is given 3 to 4 times a day and is broadcasted all over the pond. With proper feeding regime the survival rate is 75 to 80%.
However, some farmers in the tropical countries bypass the nursery phase and stock the post-larvae directly into the production ponds. The outcome in such cases is unpredictable.
(B) Grow-Out Phase:
Grow-out phase comprises grow-out phase technology, pond preparation, water quality management, stocking density, feeding technology, survival and sustainable production.
(a) Grow-out phase technology:
The culture of giant freshwater prawn can be undertaken either alone (monoculture) or in combination with various compatible fish species (mixed culture or polyculture). The location of the fish farm should be in a suitable area with warm climate and soil, with 85% water retention quality.
Rectangular perennial ponds with pollution free freshwater is required, having an area of 0.2-0.5 ha and a depth of about 1.5 m. The pond should have a sloping bottom and a sump at a deeper end for easy harvesting of the prawns.
The pond should have also an inlet pipe at the shallow end of the pond and provided with a fine sieve to prevent entry of unwanted material. An outlet should also be present at the deeper end of the pond and fitted with a fine nylon mesh screen to prevent escape of prawns.
(b) Pond preparation:
The pond should be kept dried for at least a week before filling it with water, to maintain substratum hygiene. Lime is applied at the rate of 100-200 kg/ha/ crop to maintain the desirable pH and pond hygiene. The pond is fertilised with cattle manure, (5 to 10 ton/ha), urea (100 to 150 kg/ha) and single superphosphate (200 to 300kg/ha).
Freshwater prawn is a benthic dwelling organism. They exhibit territorial behaviour and is pugnacious in nature when reared in intensive culture systems.
Therefore, increasing the surface area provides space for increased stocking and thus, reduces competition for space. Provision for shelters and artificial substrate such as branches, palm leaves, plants, horizontal framed nets, etc. when placed in ponds, not only reduces the number of aggressive encounters but also increases the survival rate of prawns, particularly in intensive culture systems.
(c) Water quality management:
The water used for grow-out operation of giant freshwater prawn (post-larvae to marketable size), should be free from toxic chemicals and pollution.
Water quality maintenance is one of the important aspects in management of prawn ponds and it should be maintained in the range given below:
(d) Stocking density:
Fifteen days after liming and fertilization, the 1-4 weeks old juveniles of prawn are stocked. In case of monoculture, the desirable stocking density is 5-8/m2 and cultured for a period of 5 to 6 months. In poly- culture systems of culture, the stocking density of prawn is reduced by 50% and along with it compatible carp fry (IMC and/exotic carps) are stocked at the rate of 300/ha.
(e) Continuous culture:
A common grow-out practice is the continuous culture, where ponds are stocked once or several times a year. They are never completely drained, except for repairs. At regular intervals harvesting is done selectively of 30 to 45 gm size of prawn for marketing.
When the whole stock is harvested, a number of small prawns will be caught which may not be acceptable in the market. So selective fishing is done with judicious management to grow undersized individuals to marketable size.
(f) Feeding technology:
M. rosenbergii in nature feeds upon a variety of food items such as aquatic worms, insects, small molluscs, crustaceans and detritus. In intensive culture, however, nutrionally balanced feed is to be provided comprising 25-30% protein in the feed. The feed is given at the rate of 20% of their body weight per day. Pellet diets have also been found to be suitable in grow-out phase.
(g) Growth and survival:
A number of factors, such as stocking density, food, water environment, are conducive of good growth and better survival rate. Prawns reared in ponds, usually reach marketable size (40-50 gm) within 5-6 months of culture.
The optimum marketable size of M. rosenbergii is about 50 gm and if allowed to grow further, survival rate would decrease drastically. The growth rate would also be slower, as much of the food would be utilised not for growth but for building of reproductive structures.
The culture period varies with the size of stocking in the pond. When stocked with post-larvae the culture period is for 6 months, while when stocked with nursery reared juveniles the culture period is reduced by 1 month. The survival rate achieved in such culture is 50-75%.
(h) Sustainable production:
Production depends on factors such as health of post-larvae, water quality, feed quality and pond management. A normal sustainable production of 1000 to 1500 kg per hectare per crop can be achieved.
However, production can be considerably increased (2500-3000 kg/hectare/crop) with provisions of pond aeration during night time (preferably between 2 to 5 a.m), exchange of water and with quality diet in the form of pellet.
The farmer through such sustainable prawn culture can achieve a high profit, much higher compared to other fish culture activities.