In this article we will discuss about the development of rabbit with the help of suitable diagrams.
Both birds and mammals have evolved from reptilian ancestors, but the developmental features of mammals have become greatly modified. The embryo develops within a specialised part of the oviduct called uterus or womb, the embryo establishing an intimate contact with the uterine wall and the young are born alive so that the eutherian mammals are viviparous.
Egg:
The egg of rabbit is 0.1 mm in diameter and is a secondary oocyte when shed from the Graafian follicle of the ovary, it is surrounded by a non-cellular, transparent striated secondary membrane, the zona pellucida secreted by the surrounding follicle cells.
Around the zona pellucida are some follicle cells (cumulus cells surrounding the oocyte) forming a corona radiata and several layers of granulosa cells from the cumulus oophorus, which breaks free from the follicle and passes with the fluid into the oviduct at the time of ovulation. In rabbit, the ovulation takes place after the copulation (i.e., female rabbit is an induced ovulator).
ADVERTISEMENTS:
The ovulation of rabbit and other mammals is found to be controlled by both the hypothalamus and pituitary gland. In rabbit, several ova mature and ovulate simultaneously. The egg is alecithal with almost no yolk, the nucleus lies in the upper half or animal hemisphere, but the lower half or vegetal hemisphere has some particles of yolk distributed evenly in the cytoplasm.
Fertilisation:
The fertilisation is internal and the egg is fertilised in the upper part of the oviduct i.e., in the fallopian tube, and it immediately undergoes a second maturation division to become a mature ovum. Two polar bodies lie within the zona pellucida near the animal pole.
In mammals, the entire spermatozoon penetrates the egg but the tail soon degenerates, penetration of sperms into an egg is brought by an enzyme hyaluronidase present in the semen. The zygote passes down the oviduct and is covered by a coat of dense albumen.
Early Embryonic Development:
Cleavage:
ADVERTISEMENTS:
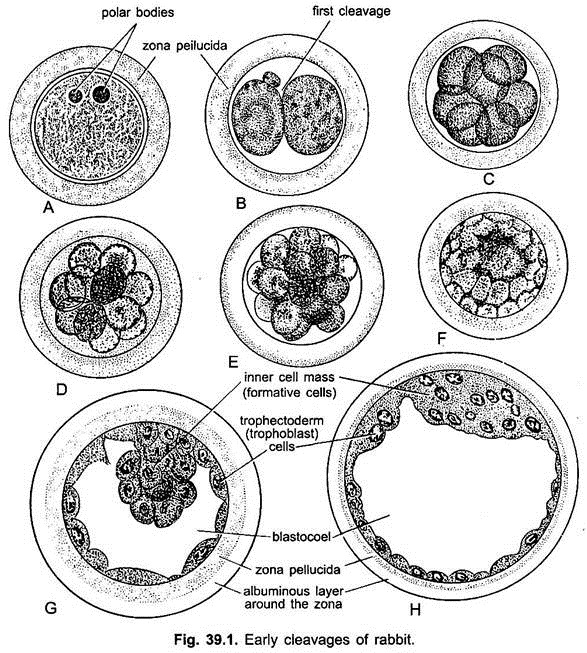
Cleavage is holoblastic but unequal. Unlike the lower animals, cleavage in mammals is not always synchronous. The number of blastomeres does not increase by a regular doubling sequence but tend to show arithmetical progression. The first cleavage furrow is vertical dividing the zygote completely into two slightly unequal blastomeres, one being smaller and more opaque than the other.
The second cleavage is also vertical and at right angle to the first, resulting into four cells. The third cleavage first occurs in two larger blastomeres and then in the remaining two smaller blastomeres, producing eight blastomeres. All the cleavages are at right angles to one another, thus, showing asynchrony of cleavage.
After this rate of mitotic division’s increases and the 16-cell stage is reached. This 16-cell stage is a solid mass of blastomeres, representing the morula. Thus, zygote, during its journey in the fallopian tube, cleaves to form the morula by the time it reaches the upper part of uterus. It lies for about 2 days free in the uterine lumen, bathed by the secretion of the uterine glands.
Soon an eccentrically placed fluid-filled cavity, the blastocoel appears within the morula which is then known as a blastocyst or blastula. The blastocoelic cavity gradually enlarges due to accumulation of the fluid that passes in the morula from the uterine cavity through the zona pellucida.
ADVERTISEMENTS:
Blastocyst:
The embryo swells rapidly and the liquid food collects in a cavity which separates an outer layer of smaller flat trophoblast cells, enclosing the fluid-filled blastocoel and the knob of round-cells, called inner cells mass, at one side of the blastocoel. The embryo is now called a blastocyst, which appears like a blastula but is not comparable to it.
Those trophoblast cells which lie above the inner cell mass are called cells of Rauber. As the blastocyst grows the inner cell mass becomes a knob-like thickening at one pole, and it is called an embryonal knob because all parts of the embryo will be derived from it.
The embryonal knob is comparable to the area pellucida of the chick, the trophoblast is equivalent to the extra-embryonic ectoderm, the cavity of the embryo to the subgerminal cavity together with the space filled by the yolk in chick, and the blastocyst is equivalent to the blastoderm of the chick.
During the first few days, it is still enclosed by the zona pellucida but it later ruptures due to the blastocyst expansion and thinning of the zona pellucida. Loss of blastocyst layer exposes the trophoblast cells to the uterine wall.
Implantation:
The trophoblast cells opposite the embryonal knob form small papillae, trophoblastic villi which penetrate into depressions or crypts in the uterine wall, so the blastocyst becomes attached or is implanted in the uterus of the mother. These villi absorb food from the uterus.
The process of implantation is initiated and controlled by a protein called blastokinin secreted by the uterus into the uterine cavity. The trophoblast cells not in contact with inner cell mass later increase in size and invade the uterine tissue, and those trophoblast cells overlying the inner cell mass continues dividing and form the extra-embryonic ectoderm (i.e., primary chorionic villi).
Inner cell mass cells facing the blastocoel become flattened and rich in rough endoplasmic reticulum. These are the primary endodermal cells, which separate from the inner cell mass and start migrating on the inner surface of the trophoblast or trophectoderm.
ADVERTISEMENTS:
The primary endoderm, thus, forms the lining of the blastocoel, now called the yolk sac. It does not seem to contribute to the definite endoderm of the embryo. As the hypoblast (or endoderm) spreads out to enclose the cavity of the yolk sac, the inner cell mass also spreads out and becomes arranged into a plate, resembling the epiblast of the blastodisc of reptiles and birds.
The blastocyst is now two cell layers thick like a gastrula, and it encloses a fluid-filled yolk sac which contains no yolk. The embryonal knob enlarges and becomes flat to form an embryonal disc which soon becomes exposed by disintegration of the cells of Rauber.
Now the epiblast becomes temporarily superficial. The blastodisc, consisting of an epiblast and a hypoblast, becomes quite sharply delimited from the remainder of the embryo. The epiblast consists of a thick plate of columnar cells, clearly distinguishable from the flat and more irregularly arranged cells of the trophoblast.
The hypoblast cells beneath the blastodisc may become cubiodal or even columnar, and thus, they differ from the cells of extra-embryonic endoderm lining the internal surface of the trophoblast.
Gastrulation:
The gastrulation of rabbit resembles with that of reptiles and birds. A primitive streak is formed, and a Hensen’s node is seen at the anterior end of the primitive streak. The primitive streak is, however, much shorter than in birds, and when fully formed, does not surpass half the length of the blastodisc, being confined to its posterior part.
The cells of the primitive streak migrate downward and sideways between the epiblast and the hypoblast. Some of the migrating cells appear to join the hypoblast and, thus, contribute to the formation of the endoderm. The loose cells migrating sideways give rise to the layer of mesoderm.
The cells migrating forward from Hensen’s node remain packed more closely and give rise to the “head process” the notochordal rudiment.
Neurulation:
After giving up the notochord and mesoderm cells (i.e., chorda-mesoderm) the remaining cells of the embryonic disc are ectoderm. Along with the separation of the mesoderm layer and the notochord rudiment, the primitive streak starts shrinking, its anterior end with Hensen’s node recedes farther and farther back.
The ectoderm of the embryonal disc during neurulation differentiates into neural ectoderm and epidermal ectoderm. The neural ectoderm lying along the centre of the disc thickens to form the neural plate, which invaginates to the neural tube. Along with this the mesoderm differentiates into dorsal epimere, middle mesomere and latero-ventral hypomere or lateral plate mesoderm.
The hypomere splits into an outer somatic mesoderm layer and an inner visceral or splanchnic mesoderm layer, enclosing the extra-embryonic coelom. These layers also form different extra-embryonic membranes and intra-embryonic parts. Like chick, the endoderm forms the primitive gut.
Development of Extra-Embryonic Membranes of Rabbit:
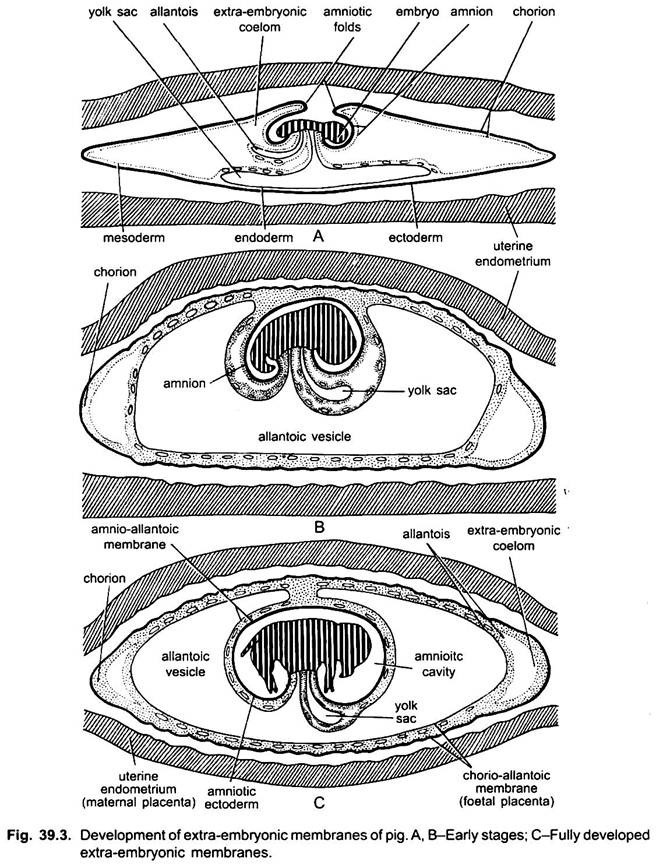
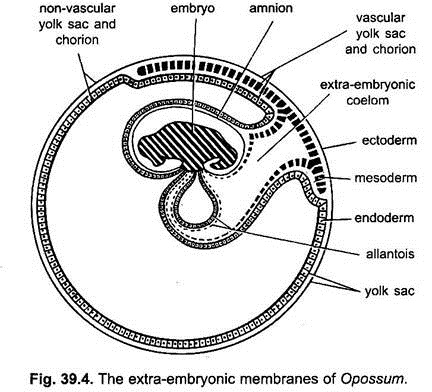
Like chick, the developing embryo of rabbit and other eutherians have four extra-embryonic membranes, namely, amnion, chorion, yolk sac and allantois. However, they differ markedly with that of chick in the method of formation and also in their morphology.
Development of Amnion and Chorion:
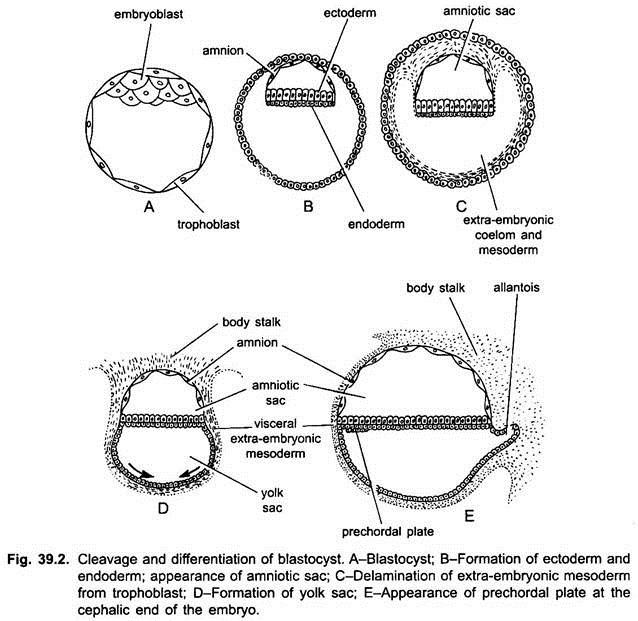
The development of amnion and chorion takes place after gastrulation and neurulation. The amnion and chorion are formed by the folds of somatopleure, called amniotic folds. The somatopleure (trophoblast or ectoderm and mesoderm) rises up behind the embryo to form a tail fold, similarly a head fold is formed later, the tail fold being larger grows more rapidly than the head fold.
Both of these folds grow over the embryo and fuse at mid-dorsal end of the embryo. The site of fusion of both of these folds is called zero-amniotic connection consisting of only ectoderm. The outer layer of these folds is the chorion which has an outer ectoderm or trophoblast and inner somatic mesoderm.
The inner layer of the folds is an amnion having an outer somatic mesoderm and inner ectoderm. Between the chorion and amnion is an extra-embryonic coelom. Thus, the embryo is enclosed in a fluid-filled amniotic cavity lined with ectoderm which acts as a protective cushion and prevents desiccation of the embryo.
Functions of Amnion and Chorion:
The amnion completely encloses the developing embryo and it contains a fluid, the amniotic fluid in which the embryo bathes. The embryo freely moves within the amnion and it also protects the embryo from abrasion and outward conclusions.
The chorion is the outer surface through which exchange of substances occurs between embryonic tissue and the maternal tissue. The chorion also develops more or less complex villi, which penetrate into the uterine tissue and, thus, greatly increase the absorptive surface of the chorion to absorb nourishment.
Development of Allantois:
The allantois of rabbit and other mammals is formed of splanchnopleure, i.e., outer endoderm and inner splanchnic mesoderm. Even before the closure of the amniotic cavity the hindgut of the embryo grows out in the form of a sac which gradually increases in the extra-embryonic coelom, i.e., space between the amnion and chorion.
It grows rapidly over the embryo outside the amnion and enlarges to form the allantois. Later on, bloods vessels develop in its wall, later some of them form the umbilical or allantoic arteries and veins. The splanchnic mesoderm of the allantois fuses with the somatic mesoderm of the chorion to form the allanto-chorion which has an outer trophoblast, a middle thickened mesoderm, and an inner endoderm.
Functions of Allantois:
The allanto-chorion is respiratory and excretory (removes CO2, urea, etc.) in function as in birds. It is also nutritive in mammals for it forms trophoblastic villi which are the major part of a placenta. It absorbs nutrient from the maternal blood.
Development of Yolk-Sac:
There is almost no yolk in mammalian eggs, yet their embryos have yolk sac. The developmental behaviour of ancestral reptilian embryos has become so strongly established that it persists even in the present-day eutherian mammals as an inheritance of their reptilian ancestry. In rabbit, the endoderm spreads over and encloses the cavity of the yolk sac.
The extra-embryonic splanchnic layer of mesoderm later on spreads over the endoderm of yolk sac. In most placental mammals, the yolk sac is small and is separated from the chorion by the extra-embryonic coelom. The embryo proper remains connected with the yolk sac by a narrow yolk stalk. During later stages, with the gradual development of allantois, the yolk sac starts reducing and finally shrivelled out.
This shrivelled yolk sac, yolk stalk and the allantoic stalk constitute the belly stalk or umbilicus. The yolk sac is nutritive in mammals also but to a much lesser degree than in the embryos developing from polylecithal eggs. But the yolk sac is never very large in mammals and it loses its nutritive function very early, then it becomes an embryonic haemopoietic organ.