In this article we will discuss about Silk:- 1. Meaning of Silk 2. Chemical Nature of Silk 3. Properties.
Meaning of Silk:
Silk is the natural protein fibre secreted by silk worm in making their cocoons and collected to make thread and fabric. According to entomologists, there are about 500 different species of wild moths spinning silk cocoons, but only few have commercial value. Here the silk produced by the larvae of mulberry and non-mulberry moths will be discussed.
However, the silk secreted by the silk worms to form the cocoon is little different from the silk thread used to weave. Because the latter lose some amount of sericin during processing. One thread of silk consists of 4-8 silk filaments and from one cocoon 1,000 yards of silk filament can be obtained.
Chemical Nature of Silk:
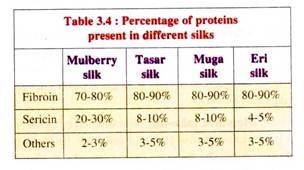
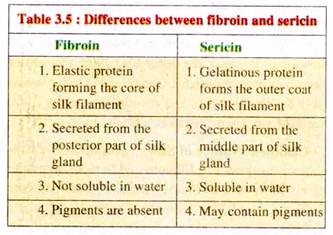
Silk principally consists of two proteins, Fibroin and Sericin, in a ratio of 80:20 in case of mulberry silk or pure silk (see Tables 3.4 and 3.5). Fibroin (C30H46 N10 O12) is a yellow coloured fibrous protein, forming the core filament of silk thread (Tables 3.4 and 3.5).
ADVERTISEMENTS:
In the silk gland it is present as a helical, soluble protein. When this liquid is pressed or extruded, its helical structure is transformed into straight, crystalline chain structure. During this transformation various amino acids are also reoriented, imparting stability to the fibre.
Fibroin is composed of 2 polypeptide chains, the heavy or H-chain (Mol. wt. 350 kD) and the light or L-chain (Mol. wt. 25 kD) which are connected with each other by disulfide bonds. Sericin, (C30H40N10D16), on the other hand, is a gelatinous protein remains as a covering around the fibroin fibre.
Being water soluble, it gets dissolved to some extent while boiling the cocoons for silk extraction. Four types of serecins are generally present in silk like Sericin-I, Sericin-II, Sericin—III and Sericin-IV.
Besides fibroin and sericin, silk also contains waxy matter, carbohydrates, minerals and some colouring substances. The golden silk contains xanthophyll, yellow silk contains caroteen and vialoxanthin, green silk has bombycin and bombycetin pigments. The colouring pigments of silk is due to the colouring pigments which were present in their food leaves.
Properties of Silk:
The ideal silk fibre should possess the following characteristics.
(A) Physical Properties:
(i) Length:
ADVERTISEMENTS:
Being the longest natural fibre, the length of an unbroken filament may be up to 1000 metres. The diameter is around 0.013 mm to 0.08 mm.
(ii) Density:
Raw silk has a density of 1.33 gm / cc and that of boiled off silk is around 1.25 gm / cc. It is lighter than cotton.
(iii) Tensile strength:
Silk is one of the strongest natural textile fibre. The tenacity of raw silk is 4.5 – 4.8 gm / denier and that of degummed silk drops to 3.3 gm/ denier. Silk can be stretched from 15 to 20% before it breaks.
(iv) Elasticity:
Silk elasticity is measured as the tension required stretching it to a definite length. It requires as high as 0.90 gm tension / denier to stretch 1%, in comparison to only 0.32 gm tension / denier required for wool.
(v) Moisture take-up:
The moisture regain is 11% in case of raw silk and only 9% following degumming. Its hygroscopic capacity is about midway between cotton and wool.
ADVERTISEMENTS:
(vi) Electric properties:
Silk is a poor conductor of electricity, though can accumulate static charge by friction. The charge can be dissipated by high humidity.
(vii) Scroop:
Scroop is the cracking sound emitted when silk is squeezed and pressed. It is not a inherent property but can be imposed by dipping it in dilute acetic or tartaric acid following by drying without rinsing.
(B) Chemical Properties:
(i) Heating:
Silk can be heated to 140°C without any decomposition. It gets disintegrated at 170°C.
(ii) Action with sunlight:
The strength of silk is reduced if exposed to UV rays of sunlight.
(iii) Action with water:
Silk can readily be wetted by water; due to swelling in water it results an increase in weight by 30-55% and diameter by 16- 18%. Boiling in water hydrolyses the fibroin more rapidly in alkaline pH.
(iv) Action with acids:
The lustre of silk increases if treated with milk acid solution. Dilute nitric acid imparts a yellow colour which cannot be washed out easily. Strong acids, on the other hand, denature its protein structure.
(v) Action with alkali:
Strong alkali dissolves the silk fibre, while mild alkaline solution may diminish its lustre.
(vi) Action with metallic salts:
During mordanting, silk can absorb some salts like stannic chloride, resulting in weight increase. But salts like zinc chloride will dissolve silk. Sodium salt has no such effect but may cause tendering, if storage for long time.